The fragrance industry is undergoing a major shift in 2026, driven by stricter global regulations. Key changes include:
- Ingredient Transparency: The EU now requires labeling for 80 allergens, up from 24. Similar rules are being adopted in Canada and the U.S.
- PFAS Bans: France, Maine, Vermont, and other regions are banning harmful chemicals like PFAS in cosmetics.
- Sustainability Standards: Companies must provide data on ingredient safety and sourcing, pushing for cleaner, safer formulations.
These regulations are reshaping how fragrances are created, marketed, and sold, with a focus on safety, ethical sourcing, and consumer trust.
Fragrance Safety Cosmetics | Regulations and Guidelines
2026 North American Regulations Affecting Fragrance Sustainability
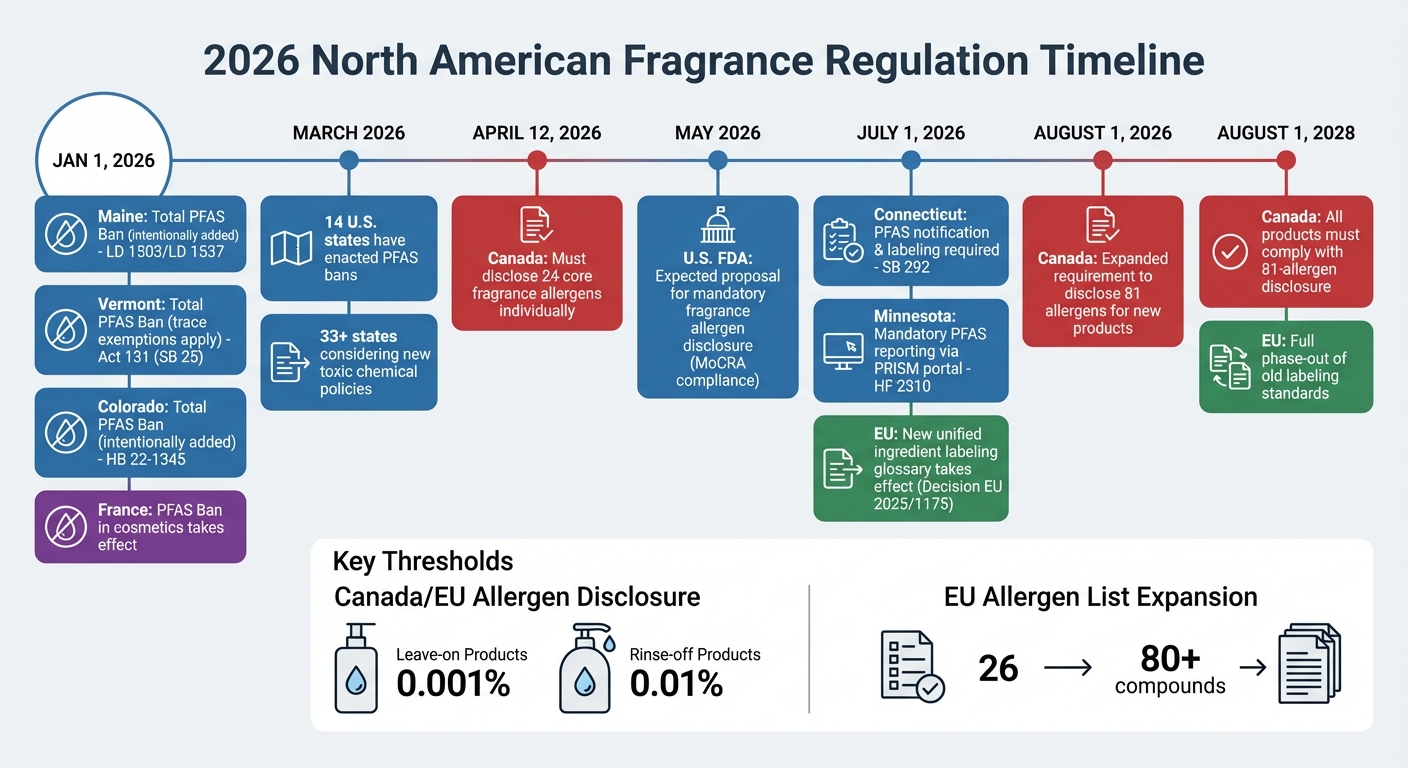
2026 North American Fragrance Regulation Timeline and State PFAS Bans
Fragrance regulations in North America are undergoing significant changes, with new rules focusing on allergen disclosure and chemical restrictions. Both the United States and Canada are introducing measures that will reshape how fragrances are formulated, labeled, and sold. These changes come with strict deadlines and penalties, pushing the industry toward greater transparency and accountability.
U.S. Fragrance Allergen Disclosure Requirements
The U.S. Food and Drug Administration (FDA) is set to propose a rule for mandatory fragrance allergen disclosure by May 2026, in compliance with the Modernization of Cosmetics Regulation Act (MoCRA). This will require brands to move away from using the generic term "fragrance" and instead disclose all ingredients at the compound level.
"Under MoCRA, the US Food and Drug Administration is required to propose a rule establishing fragrance allergen disclosure requirements for cosmetic labels." – BeautyMatter
Additionally, MoCRA mandates that brands maintain documentation to justify the safety of their fragrance ingredients. This has led many companies to collect detailed eco-toxicological data and adopt more sustainable sourcing practices.
On top of federal requirements, individual states are introducing their own chemical restrictions, particularly targeting substances like PFAS.
State — Level PFAS Chemical Bans
While federal allergen rules are still in development, state bans on per- and polyfluoroalkyl substances (PFAS) are already shaping the market. Maine and Vermont, for instance, have banned intentionally added PFAS in cosmetics starting January 1, 2026, with Connecticut following suit on July 1, 2026. By March 2026, 14 states had enacted similar bans, and over 33 states were considering new policies targeting toxic chemicals.
Companies typically avoid creating state-specific product formulations, meaning these state laws often influence national supply chains. As Cheri Peele, Director of Government and Market Policy at Toxic — Free Future, explains:
"Within the US, companies usually do not create state-specific products. When state standards change, national supply chains often change too."
Vermont’s law includes an exemption for "technically unavoidable trace quantities" from impurities or packaging migration. However, compliance requires companies to have a deep understanding of their supply chains to identify not only intentionally added PFAS but also residuals, byproducts, and processing aids.
Minnesota has gone a step further by launching its PRISM reporting portal, which requires manufacturers to report detailed chemical information for any product containing intentionally added PFAS sold in the state by July 1, 2026.
In response to these evolving regulations, fragrance manufacturers are investing heavily in new technologies and facilities. For example:
- Givaudan opened a new compounding facility in February 2026 to increase aroma chemical production capacity.
- International Flavors & Fragrances (IFF) expanded its biotechnology program in March 2026, using fermentation to create sustainable fragrance ingredients.
- Firmenich introduced AI-enabled fragrance formulation technology in January 2026, focusing on customized ingredients that meet safety standards.
| State | Effective Date | Restriction Type | Key Legislation |
|---|---|---|---|
| Maine | Jan 1, 2026 | Total Ban (Intentionally added) | LD 1503 / LD 1537 |
| Vermont | Jan 1, 2026 | Total Ban (Trace exemptions apply) | Act 131 (SB 25) |
| Colorado | Jan 1, 2026 | Total Ban (Intentionally added) | HB 22-1345 |
| Connecticut | July 1, 2026 | Notification & Labeling Required | SB 292 |
| Minnesota | July 1, 2026 | Mandatory Reporting (PRISM portal) | HF 2310 |
Canada’s New Allergen Declaration Rules
In Canada, new allergen declaration rules are setting a high bar for transparency. Starting April 12, 2026, cosmetic labels must disclose 24 core fragrance allergens individually. By August 1, 2026, the requirement expands to include 81 allergens for new products, with all products on the market needing to comply by August 1, 2028.
The thresholds for allergen disclosure are 0.001% for leave-on products like perfumes and moisturizers and 0.01% for rinse-off products such as shampoos and cleansers. Allergens must be listed in descending order of concentration, typically following the term "Fragrance (Parfum)". While Health Canada no longer requires exact concentrations of most allergens in Cosmetic Notification Forms, any substance exceeding labeling thresholds must still be disclosed.
Canada’s approach aligns closely with EU Regulation 2023/1545, which uses similar thresholds and an expanded allergen list. This alignment reflects a broader trend toward harmonized regulations across major markets.
EU and Global IFRA Standards Updates
With global regulations becoming stricter, the EU and IFRA have introduced safety benchmarks that are reshaping how fragrances are formulated and labeled across the world.
IFRA 51 Amendment: Ingredient Restrictions
The IFRA 51 Amendment updates safe-use levels for over 100 fragrance ingredients, reflecting fresh data on dermal sensitization and revised exposure models. It also introduces new phototoxicity tests, particularly for citrus-based materials, and recalculates systemic toxicity thresholds using the updated Quantitative Risk Assessment (QRA2) framework.
These changes mean that many fragrances previously deemed safe now require reformulation to meet the updated safety criteria. Compliance with these standards is essentially non-negotiable, as they are enforced through supplier agreements, retailer demands, and insurance requirements, ensuring global adherence.
The upcoming IFRA 52, set for 2025, builds on these updates without overhauling the system entirely. Alexander Mohr, PhD, President of IFRA, described the focus for 2025:
"2025 was a year of listening, learning and strengthening relationships across our global community. From advancing the IFRA Standards to engaging constructively with regulators and stakeholders, our focus has been on ensuring that fragrance remains safe, sustainable and trusted, everywhere in the world".
Meanwhile, the EU is enhancing ingredient disclosure rules with a unified labeling system.
EU Allergen and Glossary Regulations
The EU has expanded its allergen disclosure list through Regulation (EU) 2023/1545, increasing the number of declarable fragrance allergens from 26 to more than 80 compounds.
Additionally, Decision (EU) 2025/1175 introduces a unified ingredient labeling glossary, set to take effect in July 2026. Existing stock will be phased out by 2028. All new products must comply with this standard by the July 2026 deadline.
To simplify operations and prepare for similar regulations in regions like the UK and Canada, many multinational brands are adopting EU-compliant labeling as their global standard. These regulatory changes are encouraging the use of eco-conscious ingredients and transparent supply chains. As a result, manufacturers are not only streamlining compliance but also exploring sustainable production methods to align with these evolving expectations.
Your Personal Fragrance Expert Awaits
Join an exclusive community of fragrance connoisseurs. Each month, receive expertly curated selections from over 900+ brands, delivered in elegant 8ml crystal vials. Your personal fragrance journey, meticulously crafted.
Try Your First MonthHow Regulations Encourage Sustainable Ingredients and Production Methods
Tighter regulations are reshaping the way ingredients are chosen and products are made in the beauty and fragrance industries. Take France’s upcoming ban on per‐ and polyfluoroalkyl substances (PFAS) in cosmetics, set to take effect in January 2026. This move is prompting companies to replace synthetic water repellents with alternatives that are more environmentally friendly. Similarly, the EU’s expansion of allergen labeling from 24 to 80 substances, along with stricter controls on Carcinogenic, Mutagenic, and Reprotoxic (CMR) substances, is steering the industry toward safer, often plant-based ingredients. These changes are not only raising safety benchmarks but also driving innovation in sustainable production methods.
Biotechnology and circular economy strategies are gaining momentum as companies adapt to these new standards. For instance, Givaudan introduced the FiveCarbon Path™ in 2019, a program aimed at increasing the use of renewable and biodegradable carbon while incorporating upcycled carbon from industrial byproducts. These efforts aim to reduce dependency on petrochemical feedstocks. To ensure compliance and improve sustainability, companies are now using advanced assessment tools like Mane et fils’ GREEN MOTION™ and dsm-firmenich’s EcoIngredient Compass. These tools evaluate factors such as environmental impact, ingredient biodegradability, and renewable content.
Regulations are also aligning with consumer expectations for ethical sourcing and transparency. The Weitnauer Group highlights this shift:
"Modern beauty consumers demand full transparency - seeking detailed information about ingredient origins, health impacts, and ethical sourcing. Marketing claims are no longer enough".
In response, the fragrance industry is adopting initiatives focused on "field to skin" transparency. A standout example is Sana Jardin’s Orange Blossom Project in Morocco, which supports the indigenous Amazigh community by promoting sustainable practices and fair trade. The project also finds creative ways to repurpose floral waste into secondary products, ensuring nothing goes to waste.
Compliance Challenges and Industry Adaptations
Regional Regulatory Timelines and Thresholds Comparison
Fragrance brands face a maze of regional regulations, each with its own deadlines and thresholds. For example, the IFRA 51 Amendment sets a deadline of March 30, 2024, for new products and October 30, 2025, for existing formulations. Meanwhile, the EU requires allergen disclosure for new products by July 2026, with a full phase-out planned by 2028. France adds another layer of complexity with its ban on PFAS, effective January 2026.
Thresholds for ingredient disclosure also differ widely. The EU mandates disclosure at 0.001% for leave-on products and 0.01% for rinse-off products, while in the U.S., the MoCRA regulation restricts 11 specific ingredients but allows brands to list "fragrance" as a catch-all for proprietary blends . For global brands, this means the same product might require different labeling depending on the market, creating operational headaches.
These varying requirements are pushing brands to rethink their formulation and labeling strategies to stay compliant across regions.
Reformulation and Compliance Approaches
The pressure to meet stricter, and often conflicting, regulations has led brands to overhaul their ingredient sourcing and formulation processes. Traditional ingredients like rose and jasmine often exceed new allergen limits, making it challenging to maintain "natural" claims while adhering to safety standards. To address this, manufacturers are turning to advanced techniques like fermentation and enzymatic processes to create nature-identical molecules, such as lab-grown jasmine or vanilla. Additionally, selective distillation methods are being used to remove allergenic components while preserving the essence of the original scent .
When natural ingredients can’t be adjusted to meet new standards, brands are substituting them with synthetic analogs. These synthetic options mimic the desired scent profile but are designed to be more skin-friendly and less likely to cause sensitization. The financial impact of these changes is significant. The fragrance ingredients market, valued at €17.11 billion in 2024, is expected to grow to €21.94 billion by 2029, with reformulation efforts being a major driver.
Industry leaders underscore the scale of these challenges. As Elchemy highlights:
"32 new IFRA Restriction Standards to control potential dermal sensitization effects for which the systemic toxicity endpoints have also been evaluated... represent the most significant regulatory changes affecting fragrance formulation in recent years".
Some brands are taking a proactive approach by auditing their legacy formulas and adopting EU standards as their global benchmark. This strategy not only simplifies compliance but also reduces SKU complexity, making it easier to adapt to future regulatory shifts.
Conclusion
Regulations are no longer just procedural - they’re actively reshaping how fragrances are crafted, marketed, and chosen by consumers. With stricter global allergen disclosures and chemical restrictions, the fragrance industry is moving toward greater transparency and safer formulations. As the Weitnauer Group highlights:
"The tightening regulations in the global perfume and cosmetics industry are not simply regulatory hurdles - they are forces shaping the future of product innovation, go-to-market strategy, and brand value".
This shift has sparked a wave of innovation. Brands are turning to biotechnology to develop sustainable alternatives, such as lab-created fragrance ingredients like Clearwood and Dreamwood. At the same time, the market is expected to see steady growth. For consumers, this means more clarity about ingredients and safer products.
In this changing landscape, brands like Scento have a unique chance to stand out by prioritizing transparency. By offering curated 8ml decants of genuine designer fragrances, Scento allows customers to explore premium scents without worrying about hidden allergens or unsustainable components. As regulations push for detailed safety data and eco-friendly practices, nimble brands can respond faster, meeting the growing demand for openness and accountability.
Sustainability has become essential. Brands that embrace these regulatory shifts early will not only build stronger consumer trust but also secure a lasting place in a market increasingly focused on safety and environmental responsibility.
FAQs
How will the new allergen labeling rules change what I see on perfume labels?
The updated allergen labeling rules now mandate that perfume labels include a list of additional fragrance allergens when their concentrations go beyond specific thresholds. This change is designed to make it easier for consumers to spot potential allergens and choose fragrances that align with their personal sensitivities.
Will the 2026 regulations make natural fragrance ingredients harder to use?
Yes, by 2026, regulations could limit the use of natural fragrance ingredients because of tighter allergen labeling and safety requirements. This includes broader EU fragrance allergen regulations and revised IFRA guidelines, both designed to address potential sensitizers and enhance consumer protection.
What should I look for to avoid PFAS or other restricted chemicals in fragrances?
To steer clear of PFAS and other restricted chemicals in fragrances, opt for products that align with current regulatory standards. Focus on those that exclude ingredients identified as hazardous by authorities such as the EU or the US. Prioritize brands that offer clear ingredient disclosures and ensure their compliance with established safety guidelines to minimize exposure to harmful substances.







